INTRODUCTION
Tobacco use is the leading cause of preventable death (Organización Mundial de la Salud, 2019), with female smokers having a higher risk of breast cancer, cervical cancer, complications from carrying the human papillomavirus, alterations in the menstrual cycle and reproductive difficulties such as ectopic pregnancy and spontaneous abortion (Centers for Disease Control and Prevention, 2023).
In recent years, tobacco consumption in women has increased faster than in men. Moreover, treatment demand for tobacco use in women rose from 14.6% in 2001 to 30.1% in 2015 (Revuelta-Herrera et al., 2018; Reynales-Shigematsu et al., 2016). It is estimated that 9.2% (4,167,400) of female smokers in Mexico begin using tobacco at approximately 21 years of age and that they smoke an average of 6.3 cigarettes per day (Shamah-Levy et al., 2020).
The World Health Organization therefore states that governments should adopt effective measures to promote cessation and appropriate treatment for tobacco dependence (Organización Mundial de la Salud, 2005), coupled with the implementation of sex-specific control strategies.
For this reason, studies have compared the effectiveness of smoking cessation interventions in men and women. When using behavioral or cognitive-behavioral therapies combined with nicotinic and non-nicotinic therapies, women show lower rates of abstinence and, as a secondary consequence, more intense adverse symptoms than men such as nausea, vomiting, sleeping difficulties, headaches, and constipation (Castellani et al., 2020; Voci et al., 2021).
Smoking-cessation interventions designed for women have failed to achieve better results than standard interventions (Weng et al., 2021). Accordingly, variables related to the difficulty of achieving and maintaining abstinence in women have been identified, such as negative affect (Londoño et al., 2021), weight gain when quitting smoking (Kuo et al., 2022), stress (Kim et al., 2019; Martínez Leal et al., 2021) and the menstrual cycle (Wetherill et al., 2016; Zawertailo et al., 2023).
Variations in the ovarian hormones progesterone and estradiol during the stages of the menstrual cycle could have a potential effect on smoking. For example, estrogen and nicotine interact and allow an increase in the positive reinforcement obtained from smoking. Conversely, progesterone weakens the reinforcing effect of nicotine (McNulty et al., 2020; Torres & O’Dell, 2016).
In addition, in studies associating the phases of the menstrual cycle with stress in a stress-induction task, it has been observed that female smokers in the luteal phase (day 14 to 28 of the menstrual cycle) showed higher levels of cortisol than those in the follicular phase (day one to 14 of the menstrual cycle) before, during, and after the stress-induction task (Carlson et al., 2017; Montero-López et al., 2018).
In contrast, in another study by Nakajima & al’Absi, (2012), changes in stress, the desire to smoke, and withdrawal symptoms were evaluated in women who smoked tobacco and were given a stress induction task. They observed that after stress exposure, cortisol levels decreased in the luteal phase. However, no significant differences were found regarding perceived stress, the desire to smoke, or withdrawal symptoms between the luteal and follicular phases.
Ethier et al. (2023) evaluated the desire to smoke before and after an in vivo smoking session as well as the effect of a psychosocial stress task on the desire to smoke. The authors found that participants in the follicular phase had a greater desire to smoke before and after the stress induction task.
In addition, hormone levels are expected to have an effect on the tobacco-use pattern. Ethier et al. (2021) prospectively assessed the relationship between estradiol and progesterone levels and the number of cigarettes smoked. The authors evaluated smoking patterns and took urine samples from 21 women for approximately 28 days, observing more smoking when estradiol and progesterone decreased or increased together, regardless of the phase of the menstrual cycle.
To date, no consistent results have been obtained in regard to the phase of the menstrual cycle in which the most severe withdrawal symptoms and smoking cravings are experienced. The effect of withdrawal symptoms and smoking cravings on the maintenance of abstinence is therefore unclear. One limitation of the studies is that they only assessed two phases of the menstrual cycle. Other authors have recommended evaluating at least three phases of the menstrual cycle in which there are significant changes in hormone levels, which would influence smoking patterns in women (Allen et al., 2016; Schiller et al., 2014).
The objective of the present research was therefore to evaluate whether there were significant differences among the three groups at various phases of the menstrual cycle in smoking cravings and withdrawal symptoms after a stress induction task. The specific objectives were to a) evaluate whether there were significant differences in the pre- and post-test stress scores in the three phases of the menstrual cycle in regard to the base level of stress, b) assess the relationship between stress and smoking craving, symptoms of abstinence, and smoking patterns in both the pre- and post-test, and c) determine whether post-test stress predicts smoking cravings and withdrawal symptoms in the three phases of the cycle.
METHOD
Design
We used a quantitative, transversal, quasi-experimental study with pre- and post-test measurements.
Participants
A convenience probability sampling method was used. Participants were recruited from various schools and universities, as well as leading medical institutions and included 48 women aged 18 to 40 years old, resident in the State of Mexico and Mexico City. They were divided into three groups of at least 15, although seven were eliminated from the study due to their failure to complete the stress induction task, resulting in groups of 15, 14, and 12.
Inclusion criteria: women with a menstrual cycle lasting 24 to 35 days, with an average daily consumption of three cigarettes in the past 30 days, with a low risk of health complications due to tobacco use, who had at least completed elementary school.
Exclusion criteria: women who were pregnant, had recently been pregnant, used hormonal products, had a health risk associated with the use of other drugs, gynecological problems, as well as with a psychiatric disorder or premenstrual dysphoric disorder, or any other disorder related to the thyroid system, which could influence the intensity of menstruation or the presence of amenorrhea or dysmenorrhea.
Instruments and Scales
Sociodemographic questionnaire (adapted from Lira-Mandujano et al., 2012).
Fagerström Test for Nicotine Dependence (FTND) (alpha = .66) (Heatherton et al., 1991; translated and adapted by Lira-Mandujano, 2002).
- Alcohol, Smoking and Substance Involvement Screening Test (ASSIST). Screening questionnaire identifying the health risks of drug use (alpha of the tobacco subscale = .83) (Tiburcio et al., 2016).
Retrospective Baseline Consumption Questionnaire, making it possible to identify smoking patterns during the 30 days prior to the evaluation (Sobell & Sobell, 2000; adapted by Lira-Mandujano et al., 2012).
Minnesota Nicotine Withdrawal Scale (MNWS), designed to assess withdrawal symptoms with eight Likert-like items (Abrams et al., 1999; adapted by Lira-Mandujano, 2009).
Premenstrual Profile. Daily prospective self-record of the menstrual cycle. It registers the duration of the menstrual cycle and estimates the start of each phase of the menstrual cycle (Campagne & Campagne, 2007).
Premenstrual Symptoms Screening Tool (PSST). This test assesses premenstrual syndrome and premenstrual disorder according to the criteria established in the DSM-IV (alpha = .91, test-retest reliability = .56) (Steiner et al., 2003).
Questionnaire on Smoking Urges (QSU). The test assesses smoking craving through 10 items (alpha = .75) (Cox et al., 2001).
Stress Visual Analogue Scale (SVAS). This scale measures stress after exposure to an event, on a scale from 0 to 10, based on the question How much stress are you feeling right now? This scale uses a graph in a 10-cm straight line (r = .66 regarding convergent validity) (Lesage et al., 2012).
Apparatus
A laptop with a mouse was used.
Procedure
Participants were recruited through posters and social media and evaluated by phone. The sociodemographic questionnaire, FTND, and ASSIST were administered, after which they were requested to sign informed consent forms for the research.
Participants recorded their tobacco use and menstrual cycle duration on a Google Forms questionnaire for two months to identify the moment when they transitioned to each cycle phase. They were subsequently assigned to one of three groups using a simple random procedure. The groups were early follicular phase (FP1;1-3 days after the beginning of their period), late follicular phase (FP2; 10-12 days after the beginning of their period), and mid-luteal phase (LP; 21-23 days after the beginning of their period). After this, the day for the stress induction task was set.
Before the assessment day, participants were asked to refrain from smoking for eight hours. The assessment was performed in person, in a classroom with a desk, computer, and two chairs. If participants met this criterion, the QSU, MNWS, and the SVAS were administered. They then completed the Montreal Imaging Stress Task (MIST; Dedovic et al., 2005), executed using InquisitLab 6 software. The QSU, MNWS, and the SVAS tests were also administered after the task.
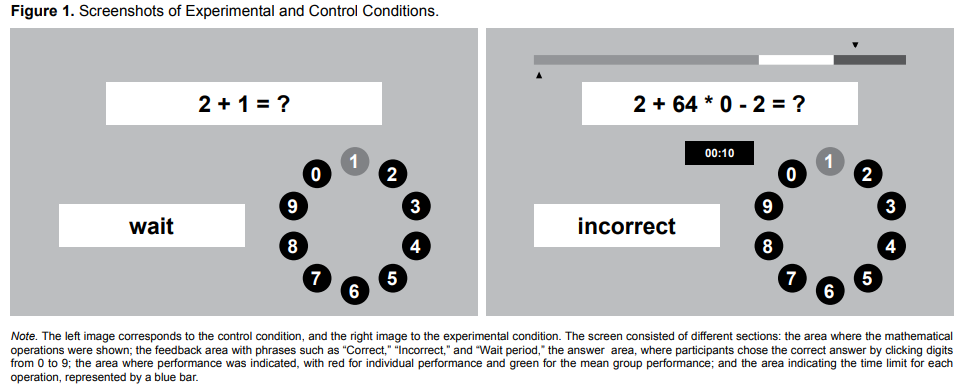
The task consisted of a brief period of practice and three conditions: test, experimental, and control, in which participants were presented with arithmetic problems with varying degrees of difficulty. During completion of the task, the researcher sat next to the participant as an observer.
The experimental condition comprised four blocks of math exercises lasting approximately three minutes each. Participants were asked to attempt to perform better than the other participants and a time limit for answering was set. In every block, participants received negative feedback: “Your performance is below average, you are not doing as well as expected, do better in the next round” (Figure 1).
Statistical analysis
We used IBM SPSS Statistics 23 for the descriptive statistical analyses. A Shapiro-Wilk normality test was undertaken in all groups (p > .05). In addition, a one-way ANOVA was carried out for the years of tobacco consumption, tobacco-use patterns, scores of risks associated with tobacco use, dependency, and premenstrual syndrome scores to determine the homogeneity of the groups.
For the main objective of this study, a one-way ANOVA was performed to compare smoking cravings and post-test withdrawal symptoms among the three groups in each phase of the menstrual cycle.
For the first specific objective, data were organized into three groups by stress level: low (0-3), medium (4-6), and high (7-10). Differences among groups were calculated using a one-way analysis of variance, as well as Sidak post hoc analyses to identify the differences within groups, using Graphpad Prisma 9.3.0 Software.
For the second specific objective, a Pearson correlation was used to assess the relationship between pre- and post-stress scores for smoking cravings, withdrawal symptoms, and smoking patterns in the 30 days prior to the assessment for each group.
Finally, for the last objective, the effect of stress on smoking cravings and withdrawal symptoms was assessed using a linear regression analysis.
Ethical considerations
This research project was reviewed and approved by the Ethics Committee of the Master’s and Doctoral Program of Psychology of the Universidad Nacional Autónoma de México, EP/PMDPSIC/010/2023.
RESULTS
Participants had a mean age of 25.07 years (SD = 6.19), had been smoking for a mean length of 5.59 years (SD = 5.52), and 56.09% were students and 24.3% employees. The remainder were completing their courses, housewives, or unemployed. In addition, 82.9% were single, 7.3% lived with their partners, and the rest were married or widowed. Most participants (85.3%) did not have children.
Mean tobacco use in the past 30 days was 3.31 (SD = 1.89) cigarettes, the mean dependency level was 2.34 (SD = 1.31; low dependency level), and the mean score on the ASSIST scale was 20.71 (SD = 6.96; moderate risk), while 53.7% of participants presented severe levels of premenstrual syndrome. Since there were no statistically significant differences among the groups, homogeneity was assumed.
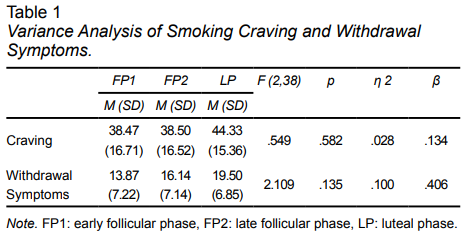
In regard to the main objective of the study, when the target variables in the three menstrual cycle phases were compared, it was found that the mean smoking cravings and withdrawal symptoms after the stress induction were higher in the LP group (Table 1). However, no significant differences were observed among the three groups (F (2,38) = 1.120, p= .353; Wilk’s lambda = .889, partial eta squared = .057, Table 1).
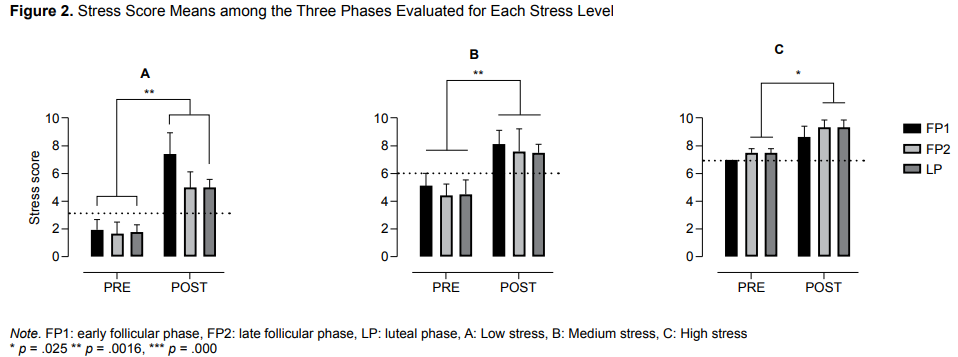
An analysis to determine whether there were significant differences in mean pre-and post-test stress levels in the three phases of the menstrual cycle with respect to the basal level of stress found that the LP group presented a higher mean stress score than the others both before (M = 8) and after the stress induction task (M = 9.3). In the low and moderate stress levels, group FP1 scored the highest averages in both the pre-test and the post-test. However, group LP scored the highest overall means in the high stress level in both the pre- and post-test.
Significant differences were found among groups (Figure 2) in low (F (1, 15) = 34.74, p < .0001), medium (F (1, 15) = 75.85, p < .0001), and high stress levels (F (1, 8) = 31.38, p = .0005). Using Sidak’s multiple comparisons, significant differences were observed in the low stress level between groups FP1 and LP [CI = 95%; -8.8, -2.1 and -5.2, -1.2 respectively], in the medium stress level among the three groups FP1, FP2, and LP [CI = 95%; -4.4, -1.5; -4.5, -1.6 and -4.9, -1.0 respectively], and in the high stress level between groups FP2 and LP [CI = 95%; -3.2, .0 and -3.2, -.2].
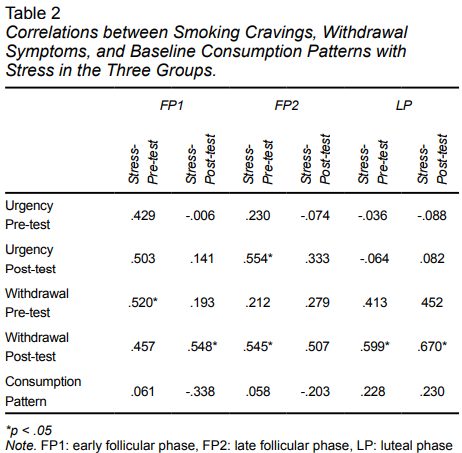
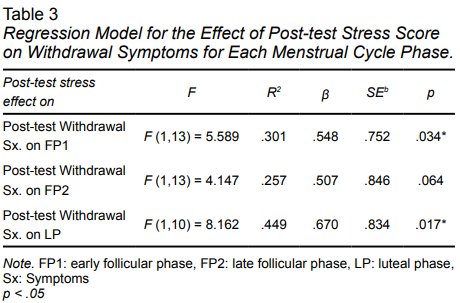
In regard to the relationship between stress and withdrawal symptoms, as well as between stress and smoking cravings in the pre- and post-tests in the three menstrual cycle phases (Table 2), there was a moderate correlation between stress and smoking cravings in the pre-test with group FP2, with withdrawal symptoms in the pre-test scores of FP1, and withdrawal symptoms in the post-test scores in groups FP1 and LP. There was a significant correlation with withdrawal symptoms in the post-test scores for groups FP1 and LP (Table 3). Correlations between the consumption pattern in the baseline with pre- and post-test scores were also assessed, with no significant correlations being found in any group.
As for the third objective of this study, a simple linear regression model with the input method was calculated (Table 3) to predict the effect of post-test scores on post-test withdrawal symptoms in the three menstrual cycle phases. The regression equation was statistically significant for group FP1, F (1, 13) = 5.589, p < .005, with R2 = .30, indicating that 30% of the change in withdrawal symptoms can be explained by the post-test stress scores. The regression equation was -.229 + 1.779 * (post-test stress score). A significant regression was also found for group LP, F (1,10) = 8.162, p < .001, R2 = .44. The regression equation was 2.621 + 2.383 * (post-test stress score).
DISCUSSION AND CONCLUSION
The main objective of this study was to assess the differences between smoking craving and withdrawal symptoms in three groups at different phases of their menstrual cycle, after they had been given a stress induction task. The results showed no differences in the dependent variables of the study among groups.
The results of the present study are consistent with studies that have observed no differences in smoking cravings or withdrawal symptoms among the phases of the menstrual cycle (Allen et al., 2013;2014), which have concluded that women experience symptoms that hamper the long-term maintenance of abstinence in all phases of the menstrual cycle.
Allen et al. (2014) found no significant differences in these variables. Conversely, they observed more withdrawal symptoms and perceived stress during the luteal phase in women in acute abstinence. Carlson et al. (2017) observed an increase in the desire to smoke during the luteal phase in women who had participated in a stress induction task.
For the first specific objective, it was found that in the low and medium stress levels, the FP1 group showed the significantly highest mean of stress, whereas in the high stress level, the LP group had a significantly higher mean. After a stress induction task during various phases of the menstrual cycle, Montero-López et al. (2018) found that there was more stress during the luteal phase, given that higher cortisol levels were observed after the stress induction task.
Several studies have shown a correlation between cortisol production and premenstrual symptoms, suggesting that increased activity in the hypothalamic-pituitary-adrenal axis predicts the severity of premenstrual syndrome (Huang et al., 2015). Valdez-Piña et al. (2024) demonstrated that perceived stress significantly predicts smoking patterns in women although not in men, concluding that smoking relapses in women are closely linked to mood variables.
In the second objective, an analysis of the relationship between stress and the urge to smoke as well as withdrawal symptoms in the pre-test and post-test showed that there was a correlation between withdrawal symptoms and stress during the pre-test or the post-test measurements in all groups. Nonetheless, in group LP, there was a correlation between withdrawal symptoms and pre- and post-test stress scores, with post-test scores showing a greater correlation.
In regard to the previous results, the original hypothesis was that withdrawal symptoms would only correlate with the stress scores in the LP group due to the presence of premenstrual syndrome. However, stress had a similar effect on the three groups, with a greater effect on the LP group, as borne out by the correlations observed in the pre- and post-test stress scores in this group. This result is consistent with other studies in which stress has correlated with tobacco withdrawal symptoms, suggesting that premenstrual syndrome can increase the intensity of withdrawal symptoms (Cross et al., 2017; Montero-López et al., 2018).
Thirdly, in the regression analysis, in the FP1 and LP groups, post-test stress scores significantly predicted post-test withdrawal symptoms. This may be due to women suffering from premenstrual syndrome in the LP and similar intensity symptoms experienced during the days associated with FP1, which could affect their smoking behavior (Lovick et al., 2017; Núñez-Lauriano et al., 2023).
Conversely, Saladin et al. (2015) showed that an increase in progesterone during the luteal phase has a protective effect on the desire to smoke. This has been taken as an indication of the importance of progesterone in the context of abstinence, despite it having less impact on emotional regulation during everyday tobacco consumption.
These results suggest that, in the context of smoking cessation interventions, during the LP and the FP1, women should implement coping and stress-management skills to facilitate the long-term maintenance of abstinence (Martínez Leal et al., 2021). Furthermore, since women experienced the lowest stress scores during FP2, stress-management training during this phase would presumably be more effective in creating significant changes.
Bearing this in mind, and in keeping with the goal of selecting a day to quit smoking (Organización Panamericana de la Salud [OPS], 2020), smoking cessation should commence before the luteal phase, when premenstrual syndrome occurs, after women have been taught stress-management techniques. This study also demonstrates the viability of the menstrual cycle record as an alternative for biological markers, which could make it easier for women to identify the duration of their cycles and the times when increased discomfort is most likely.
Limitations of this study include the fact that biological markers were not used to identify the menstrual cycle phase or cortisol levels. There was no restriction on the use of painkillers, and the time when participants were given the stress induction task varied.
Moreover, according to the results of the ASSIST test, the sample had a moderate health risk regarding their tobacco consumption, which could have affected the intensity of their withdrawal symptoms, probably reducing their effect. Given this characteristic, it may only be possible to generalize these results to samples with these traits. In the case of Mexico, 82.3% of regular smokers smoke 7.4 cigarettes a day (Paz-Ballesteros, et al., 2019).
Furthermore, it is important to note that in this study, comparisons were made among groups to guarantee the homogeneity of tobacco consumption patterns, dependency levels, and premenstrual symptoms in each of the three menstrual cycle phases. With these variables in mind, a simple randomization of the groups was performed. Nevertheless, future research could explore block randomization or intragroup comparisons.
At the same time, given the possible effect of the sample size on the lack of significant differences in smoking patterns, dependency levels, and premenstrual syndrome symptoms among the three phases of the menstrual cycle, in further studies it could be useful to increase sample size and identify more of the characteristics of participants in the sampling methods used.
In future research, these results should be compared with those of women with high dependency levels, in the menopause or perimenopause, and to assess the relationship between stress and anxiety and depression at different phases of the menstrual cycle using a stress induction task. Moreover, in interventions designed to help women quit smoking (Allen et al., 2022; Nair et al., 2020), clearly defining the dates when an abstinence period started and assessing the effect of this on the various phases of the menstrual cycle phases could be valuable.
In conclusion, smoking cravings and withdrawal symptoms did not differ among the phases of the menstrual cycle. Nonetheless, the stress experienced by participants after a stress induction task was greater during the FP1 and LP, which had an effect on withdrawal symptoms, mainly in the LP group with women with high stress levels. Women would therefore benefit from implementing stress-management strategies during the FP1 and LP.