INTRODUCTION
The term “schizoaffective psychosis” was introduced by Jacob Kasanin to describe a group of patients with both affective and psychotic symptoms similar to schizophrenia, with acute onset in youth, following a stressful life event and with sufficient premorbid social adjustment. The term was further refined by Schneider in his famous “cases in the middle” as chronic psychoses behaving differently from schizophrenia. However, there has been controversy regarding the existence of the disorder as a distinct nosological entity (Arenas & Rogelis, 2006) ever since the term was first coined. It has been argued that it should be classified as a variant of schizophrenia or a major affective disorder (Abrams et al., 2008; Lake & Hurwitz, 2006), an in-between category (Cheniaux et al., 2008), or an independent entity (Tsuang, 1991). It has therefore been considered a clear failure of psychiatric disease classification systems (Shaw et al., 2008).
The fifth version of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) defines schizoaffective disorder as a disorder with a prolonged major affective disorder as well as meeting criterion A for schizophrenia. At least two weeks of hallucinations and/or delusions without affective symptoms are also required (American Psychiatric Association, 2013).Conversely, the ICD-11 (International Classification of Diseases, 2024) defines it as the presence of all the characteristics of schizophrenia concurrently, or within a few days, with affective symptoms for at least four weeks. The main difference between these classification systems is that the ICD-11 conceptualizes schizoaffective disorder as a cross-sectional diagnosis, whereas the DSM-5 regards it as a longitudinal condition (Peterson et al., 2019). This complicates the establishment of an accurate diagnosis, which would facilitate the treatment, course, and prognosis of the condition.
This longstanding nosological debate is mainly due to the overlap between the clinical manifestations of schizoaffective disorder and those of schizophrenia and bipolar disorder, combined with the lack of specific biological markers to support diagnosis. Attempts have been made to pinpoint unique characteristics to identify schizoaffective disorder. For example, it has been found to be more prevalent in women than men, and it has an earlier age of onset, and a better prognosis and premorbid adjustment than schizophrenia (Benabarre et al., 2001; Saracco-Alvarez et al., 2009). Processing speed has also been proposed as a differentiating marker, being significantly higher in individuals with schizoaffective disorder than in those with schizophrenia (Mondragón-Maya et al., 2019). However, other studies have concluded that schizoaffective disorder follows a similar pattern to that of schizophrenia and bipolar disorder, with the same degree of deficient cognitive functions (Amann et al., 2012; Hartman et al., 2019).
Brain imaging techniques could be a source of specific biomarkers for psychiatric disorders. There is dearth of studies using computed tomography (CT) or magnetic resonance imaging (MRI) to analyze brain structures in schizophrenia and schizoaffective disorder. Most of them (Kuo & Pogue-Geile, 2019) fail to provide clear evidence of differences between diagnoses because the two pathologies are commonly included in a same group study or compared with other disorders. For example, Amann (2016) evaluated structural changes using voxel-based morphometry across four study groups with 45 subjects each: schizoaffective disorder, schizophrenia, bipolar disorder, and healthy controls. Patients with schizoaffective disorder were found to have a generalized reduction of gray matter similar to that of the schizophrenia group, and in both groups, this reduction was greater than in subjects with bipolar disorder Getz et al., 2002. Similar findings were observed in the study by Ivleva (2013). One hundred and forty-six patients with schizophrenia and 90 with schizoaffective disorder had gray matter reductions in similar regions compared to 200 healthy controls, while 115 patients with bipolar disorder had reductions in more limited regions. By including bipolar disorder, these studies concluded that schizoaffective disorder is closer to schizophrenia than bipolar disorder.
There is a need for research focusing exclusively on comparing the neuroanatomy of schizophrenia and schizoaffective disorder. Understanding the unique brain changes of each disorder can shed light on the clinical heterogeneity observed in practice, which can have significant implications for classification and clinical management. The main objective of this study is to compare total intracranial volume (Kijonka et al., 2020), specifically gray matter (GM), white matter (WM), and cerebrospinal fluid (CSF) volumes, in subjects diagnosed with schizophrenia and schizoaffective disorder. To this end, we used an MRI database drawn up by the Ramón de la Fuente Muñiz National Institute of Psychiatry (INPRFM).
METHOD
Study design
We performed an observational study with comparison groups, involving secondary analysis of a brain image database of subjects with schizophrenia and schizoaffective disorder.
Study sample
This study retrospectively used voxel-based morphometry on brain MRI images from the INPRFM database. All cases with a diagnosis of schizophrenia and schizoaffective disorder in the hospital registry were included, followed by a search for those who had undergone magnetic resonance imaging in the past five years. Since clinical files were not documented, only images with subjects’ sex and diagnosis were recorded. The final sample size was 55 complete images for each study group: subjects diagnosed with schizophrenia and those diagnosed with schizoaffective disorder. Inclusion criteria were brain MRI images of subjects evaluated at the INPRFM with a diagnosis of schizophrenia or schizoaffective disorder according to DSM-5-TR criteria, with at least two continuous years of this diagnosis, and images including a complete protocol of the scanner’s routine sequences. Exclusion criteria were brain images with anything distorting any sequence and preventing the measurement of the volume to be studied, and images of subjects with structural diseases or anatomical variables. This study was approved by the INPRFM ethics and research committee.
MRI Acquisition and processing
Brain images were acquired with a Philips Ingenia 3 Tesla scanner and subjected to scanner protocol sequencing with a T1-weighted spin-echo sequence. The 110 brain images from the INPRFM database were processed using the method described earlier. Images were converted from the DICOM Connectivity Framework format to NII (MITA, 2020). Normalization was performed using Statistical Parametric Mapping 12 (SPM12) software in MATLAB (MathWorks, 2024) to remove artifacts and/or adjust the temporal and dimensional space of the images. This process corrects image deformities, matching the image in the reference field that adapts the original image, creating a lack of uniformity. The image must be normalized, and processing must be carried out using software that evaluates and adjusts distortions as part of the analysis, to analyze brain images using reference images (CAT12, 2023; The FIL Methods Group, 2014).
The procedure involves iteratively performing tissue classification by estimating tissue posterior probability mappings (TPMs) from the functional/anatomical image intensity values of the reference and registration, estimating the nonlinear spatial transformation that best approximates the posterior and anterior TPMs until they converge. Once the images had been normalized, they were subjected to a segmentation algorithm into classes according to the tissues observed: gray matter, white matter, and cerebrospinal fluid using the Computational Anatomy Toolbox CAT12 (Kurth, Gaser & Luders, 2015). Areas not belonging to the tissues of interest were removed.
The segmented images were 121 x 145 x 121 voxels with a voxel size of 1.5 x 1.5 x 1.5 x 1.5 mm. The range of values was varied so that each voxel had a value of approximately -800 to 32,800. At this point, images from the schizoaffective disorder group were automatically normalized in intensity to the same range of values as the schizophrenia group. Areas containing bone and adjacent soft tissue were removed. Finally, the volumetric study was performed using CAT12 (CAT12, 2023) for each of the 110 brain images, and the total volumes of GM, WM and CSF were measured.
Statistical analysis
For statistical analysis, we used SPSS Statistics 21. For demographic variables, age and sex, we used descriptive statistics, frequencies and percentages for nominal variables and means, and standard deviations for continuous variables. Initially, sex and age were compared between both groups using Chi-square (χ2) and Student’s t-test for independent samples, respectively, to determine their inclusion as covariates to control in subsequent comparative analyses. As we observed a difference by sex between groups, this variable was included as a covariate in subsequent analyses.
To obtain the total intracranial volume (TIV) in cubic centimeters (cc), the three values of interest, WM, GM and CSF (Kijonka et al., 2020), were added together and the meninges were not included in the analysis. General linear models were used to compare these variables between the schizophrenia and schizoaffective groups. These included the group (schizophrenia versus schizoaffective) and sex (men versus women) as fixed factors, and group by sex interaction.
Ethical considerations
The study protocol and procedures were approved by the Institutional Review Board of the INPRFM (CEI/C/062/2022), a specialized psychiatric institution dedicated toto clinical care, research, and teaching. Allimages were obtained from the INPRFM brain imaging department database.
RESULTS
Of the total 110 images obtained, 44.5% (n = 49) corresponded to men and the remaining 55.5% (n = 61) to women. Differences by sex were observed, where 67.3% (n = 37) of the images in the schizoaffective group corresponded to women, compared to 43.6% (n = 24) of the images in the schizophrenia group (χ2 = 6.2, 1 gl, p = .01). The average age for the schizophrenia group was 37.6 years (SD 16.82), and 40.20 years (SD 10.14) for the schizoaffective group, with no differences between groups (t -.9, 108 df, p = .33).
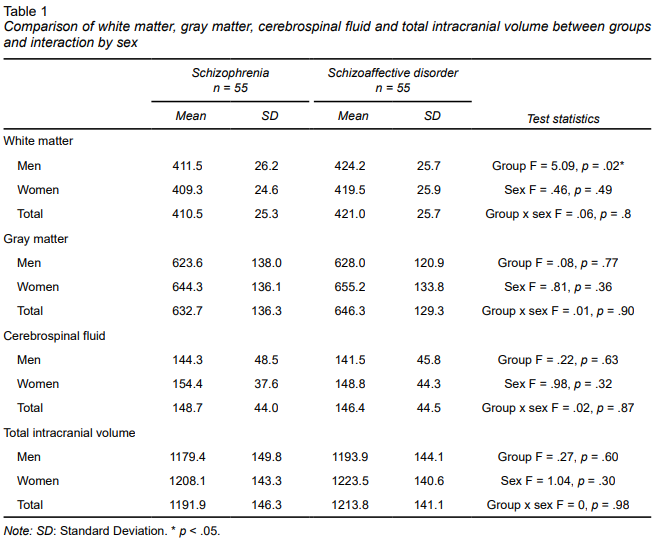
The results of the comparison of TIV are shown in Table 1. A difference was observed between the schizophrenia and schizoaffective groups in WM volume, which was greater in the schizoaffective group. This difference was not observed between men and women, nor was there any interaction between group and sex. Regarding GM, CSF, and TIV, no differences were observed between groups, by sex, or in the interaction between group and sex.
DISCUSSION AND CONCLUSION
In this study, we compared the total intracranial volume of subjects diagnosed with schizophrenia and schizoaffective disorder. A significant difference was found in WM volume between the two groups, whereas TIC, GM and CSF showed no differences. These findings may support the conceptualization of schizoaffective disorder as a separate condition from schizophrenia. However, there is still limited evidence from brain imaging comparing structural brain changes exclusively between these study groups. These diagnoses are usually classified as a single group in studies comparing them with other conditions such as bipolar disorder and depression (Ivleva et al., 2012; Madre et al., 2013; 2016). This strategy does not allow us to pinpoint specific differences and similarities in brain structures between schizoaffective disorder and other disorders, particularly with schizophrenia, limiting the evidence that could clarify part of the nosological difficulty explained.
GM volume changes are the most commonly reported findings in current research. Amann et al. (2016) and Ivleva et al. (2013) concluded that GM reductions in individuals with schizoaffective disorder resemble those in individuals with schizophrenia when compared to bipolar disorder. This could correlate with our finding of no difference in GM in the comparison between schizoaffective disorder and schizophrenia.
Differences between schizophrenia and controls show a greater reduction in total brain volume (TBV), including GM and WM, as described in many samples (Bethlehem et al., 2022; Fusar-Poli et al., 2013). However, these studies do not specifically analyze schizoaffective disorder as we did with the sample in our study.
The changes observed in WM have only been explored in patients with schizophrenia. Kubicki et al. (2005) included abnormalities in WM through structural MRI, voxel-based morphometry, and diffusion tensor imaging. Their findings suggested an alteration in the distribution of white matter cells and in the processes of maturation and myelination in individuals with schizophrenia. A study comparing monozygotic and dizygotic twin pairs discordant for schizophrenia with healthy control twin pairs found that both schizophrenic twins and their healthy twins had a decrease in total WM volumes. However a decrease in GM was only observed in the schizophrenia group (Hulshoff Pol et al., 2004). These authors concluded that WM alterations reflect a higher genetic risk. GM changes, however, are more associated with environmental factors, suggesting that the findings in our study are due to differences in brain development origin, with fewer alterations due to environmental influences in the schizoaffective group.
Our study compared the structures mentioned above, highlighting that the group with schizoaffective disorder has a larger WM volume than the schizophrenia group. However, it is striking that, in other studies, the total volumes of both groups are smaller than the average of healthy control groups. For instance, in schizophrenia, a reduction of over 2% was estimated (Haijma et al., 2013), which in our sample increases the general volume reduction percentage in individuals with schizophrenia to 8.3% and in those with schizoaffective disorder to 6.6%. This is above the average of other studies (1,300 cc), which can be explained by the progression of volume loss with advancing age, as our sample is in the fifth decade of life. These differences could explain the various clinical presentations existing between these disorders. This finding underscores the importance of continuing to investigate differences between the two groups to identify the specific characteristics of each disorder, define possible biological markers, and increase the evidence of schizoaffective disorder as distinct from schizophrenia.
Although the use of whole brain volumes does not identify local anatomical abnormalities, it does indicate the presence of a general process. More advanced technologies could provide a more detailed analysis of these neuroanatomical changes. Some studies have used functional MRI (fMRI) to examine patterns in brain function and propose phenotypes or biomarkers for psychiatric disorders (Miranda et al., 2021). For psychotic disorders, Yan et al. (2022) used fMRI time series to integrate neural networks, clustering and interpretation for schizophrenia, bipolar disorder with psychotic symptoms, schizoaffective disorder and healthy subjects. This study proposed a deep classification and clustering framework to interpret disease-related networks and visualize the relationships among these groups. Interestingly, even with newer approaches, the schizoaffective group proved the most challenging to isolate and classify.
The following limitations should be considered. When examining WM, GM, and CSF volumes, only a general idea of structural changes can be obtained, making it difficult to correlate more directly with clinical manifestations of the conditions and possible hypotheses about their pathophysiology. Healthy controls were not considered in this comparative analysis, which may have biased the interpretation of results. Additionally, clinical information (time since diagnosis and pharmacological treatment) was unavailable for this study. Both variables could have implications for the structural changes found, as observed in schizophrenia (Moncrieff & Leo, 2010; Yang et al., 2021; Andreasen et al., 2013).
In conclusion, this study provides evidence on the neuroanatomical differences between schizoaffective disorder and schizophrenia, supporting the notion that schizoaffective disorder is not merely a variant of schizophrenia, but a clinically and biologically distinct entity. Our findings emphasize the importance of research focusing exclusively on comparing the neuroanatomy of these disorders for a deeper understanding of their underlying pathophysiology. Additionally, they draw attention to the need to consider these differences in clinical practice, which could have significant implications for the diagnosis, nosological classification, and therapeutic management of patients with schizoaffective disorder.
Future research using functional brain MRI could shed further light on the differences in the clinical phenomenon in patients and contribute to the development of a more consistent nosology of psychosis.